(c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube.
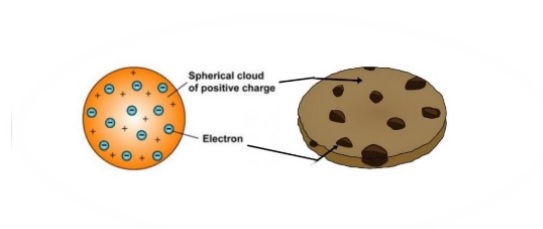
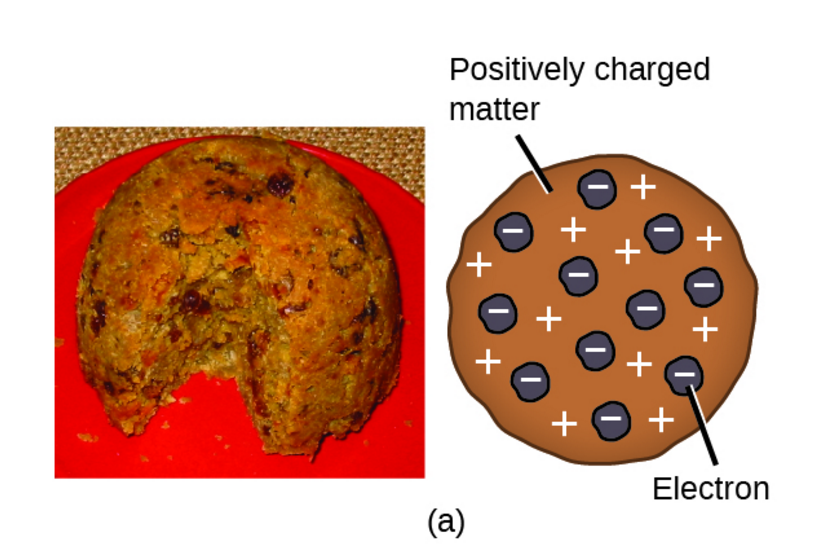
(b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. He could then calculate the charge-to-mass ratio (e/m) of the electron. The magnetic field deflected the electrons into circular paths of known radius (r). In 1904, Thomson proposed the “plum pudding” model of atoms, which described a positively charged mass with an equal amount of negative charge in the form of electrons embedded in it, since all atoms are electrically neutral. Thomson produced a visible beam in a cathode ray tube. Thomson used an electric field (V) to accelerate electrons into a magnetic field (B). The plum pudding model is one of several historical scientific models of the atom.First proposed by J. However, the positively charged part of an atom was not yet well understood. The plum pudding model of the atom The current model of the sub-atomic structure involves a dense nucleus surrounded by a probabilistic 'cloud' of electrons. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative, subatomic particles-the electrons-were known. The modern atomic theory establishes the concepts of atoms and how they compose matter.
